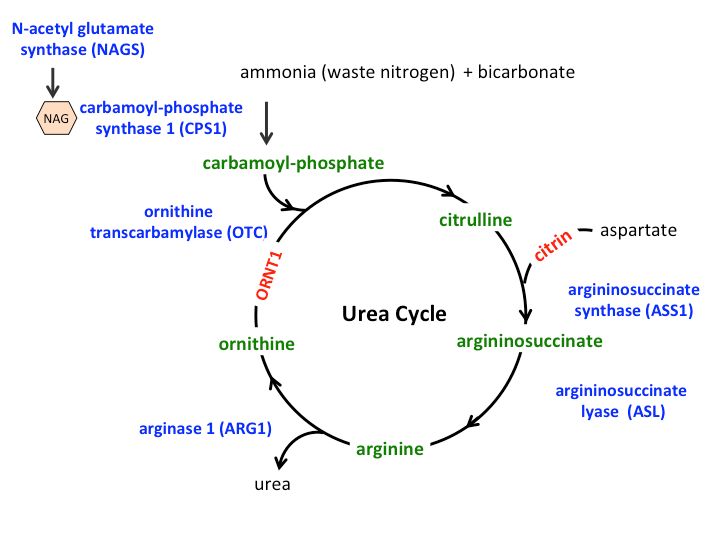
The urea cycle disorder treatment market comprises products that are used for the management and treatment of urea cycle disorders. Urea cycle disorders are a group of rare genetic disorders that affect the body’s ability to remove ammonia from the bloodstream. Common symptoms of urea cycle disorders include lethargy, poor appetite, vomiting, and seizures. Left untreated, an excess of ammonia in the bloodstream can lead to brain damage, coma, or death. The global prevalence of urea cycle disorders is estimated to be around 1 in 35,000 people. Effective treatment consists of medications to reduce ammonia production, a low-protein diet, supplementation with essential amino acids and carnitine, ammonia scavengers, and sometimes liver or kidney transplantation.
The global U.S. urea cycle disorders treatment market is estimated to be valued at US$ 587.52 million in 2024 and is expected to exhibit a CAGR of 5.3% over the forecast period 2024-2031, as highlighted in a new report published by Coherent Market Insights.
Market key trends:
One of the key trends in the urea cycle disorders treatment market is the growing adoption of newborn screening programs. All 50 U.S. states currently require newborn screening for various conditions, including several urea cycle disorders. Early diagnosis through newborn screening allows for prompt treatment before any signs or symptoms appear, which significantly improves long-term health outcomes. As public awareness about newborn screening increases and screening panels continue to expand the number of conditions covered, the adoption rate of newborn screening is expected to rise steadily over the forecast period. This will identify more cases of urea cycle disorders early on and drive market growth.
Porter’s Analysis
Threat of new entrants: The threat of new entrants in the U.S. urea cycle disorders treatment market is moderate. Establishing manufacturing facilities and gaining regulatory approvals is capital intensive which restricts entry of new players.
Bargaining power of buyers: The bargaining power of buyers in the U.S. urea cycle disorders treatment market is low as the diseases have no alternative treatment options and patients have to rely on the available treatments.
Bargaining power of suppliers: The bargaining power of suppliers is moderate as there are few suppliers for raw materials and manufacturing equipment required for production of urea cycle disorders treatment drugs and devices.
Threat of new substitutes: There exist no close substitutes for drugs treating urea cycle disorders limiting the threat from substitutes.
Competitive rivalry: The competitive rivalry in the market is high owing to presence of large as well as small players offering similar treatment options.
Key Takeaways
The Global U.S. Urea Cycle Disorders Treatment Market Size is expected to witness high growth over the forecast period. Being a rare disease with limited treatment options, demand for drugs and devices remains strong. The global U.S. urea cycle disorders treatment market is estimated to be valued at US$ 587.52 million in 2024 and is expected to exhibit a CAGR of 5.3% over the forecast period 2024-2031.
Regional analysis: The Western and Midwestern regions hold around 60% share of the U.S. Urea Cycle Disorders Treatment market driven by higher diagnosed patient base and healthcare expenditure. States like California, Texas and New York are major markets.
Key players: Key players operating in the U.S. Urea Cycle Disorders Treatment market are Kawasumi Laboratories Inc., Blood Purification Technologies Inc., AWAK Technologies Pte. Ltd, Triomed AB, Asahi Kasei Medical Vo. Ltd, US Kidney Research Corporation, Merit Medical Systems, NIPRO Medical Corporation, Fresenius SE & Co. KGaA. Kawasumi Laboratories Inc. and Blood Purification Technologies Inc. collectively hold over 30% market share.
*Note:
1. Source: Coherent Market Insights, Public sources, Desk research
2. We have leveraged AI tools to mine information and compile it